SOLVED: and 4,300 m. Assume that oxygen is 21% by volume in the atmosphere. At sea level, the atmospheric pressure is 760 mm Hg = atm 101 kPa. The change in pressure

SOLVED: The pressure 35.( mcters under water is 445 kPa. Conversion fectors 1kPa 9.869 atm atm 780 mmHg Whal is this pressure in atm? pressure: What this pressure in mmHg? mmdg pressurc:

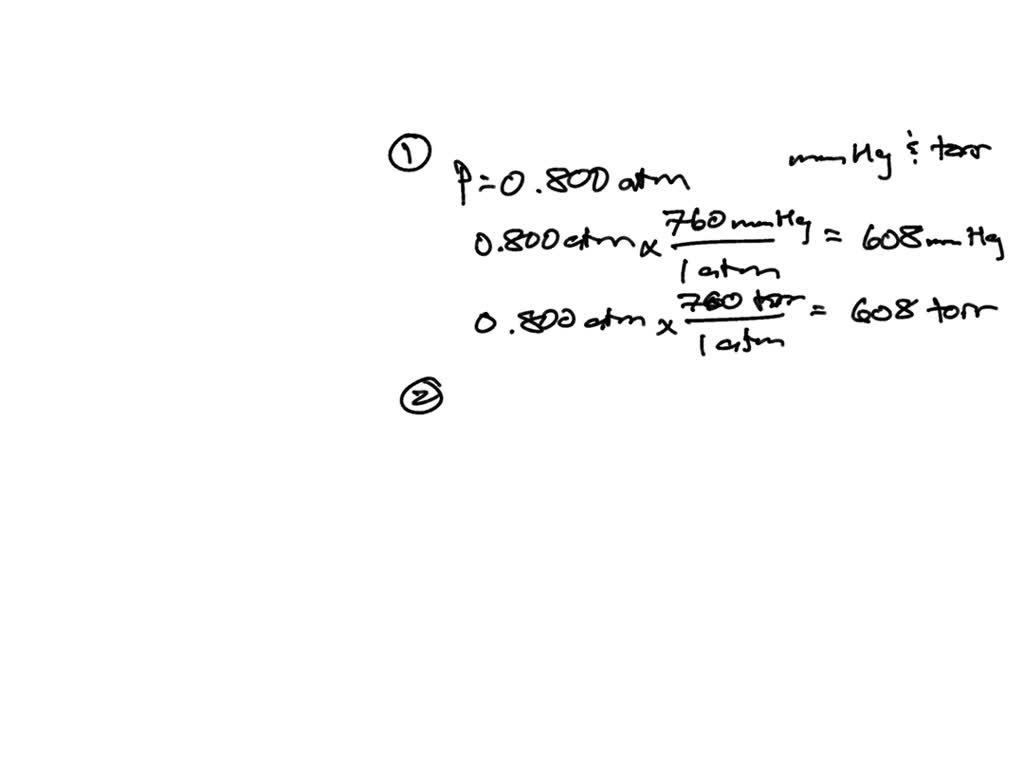
SOLVED: QUESTION 1: The pressure 80.0 meters under water is 886 kPa. Conversion factors 1 kPa=9.869×10−3 atm 1 atm=760 mmHg What is this pressure in atm? pressure: atm What is this pressure

Amazon.com: WAR211A.BA0782 Watch Tag Heuer Men's Carrera Stainless steel case, Stainless steel bracelet, Black dial, Automatic movement, Scratch resistant sapphire, Water resistant up to 10 ATM - 100 meters - 330 feet :
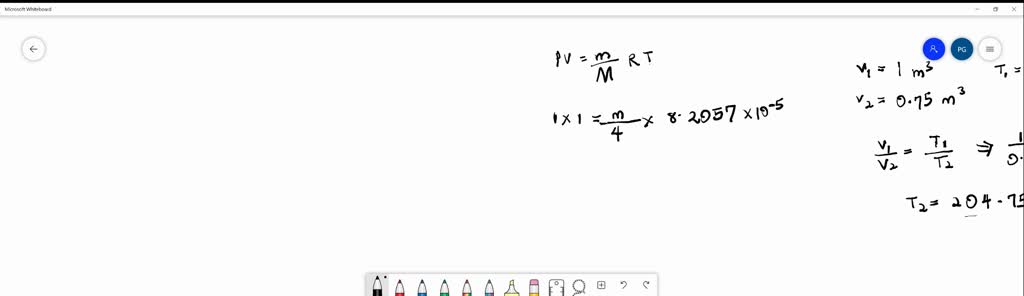
SOLVED: A cubic meter of helium originally at 0-degrees C and 1-atm pressure is cooled at constant pressure until the volume is 0.75 𝑚^3.. How much heat in kcal is removed? CvHelium =

SOLVED: please answer fast. 1. Water is flowing in a 4 in Schedule 40 steel pipe at 20C and 1 atm. Orifice meter with orifice diameter 6.2 cm is fitted in pipe